We discovered a new group of porous coordination polymers (MOFs). These materials may find use in preservation and separation of gases or as effective sorbents of organic pollutants.
Until now, MOFs had been prepared using linkers carrying carboxylic acids (RCOOH), phosphonic acids (RPO2H2), or N-donor groups. Compared to the linkers with carboxylic acids used until now, phosphinic acids bind more firmly to metal centres. At the same time, they do not impair this structure like other ligands do, e.g. those containing phosphane groups. Our new method allows for controlling hydrophilicity / hydrophobicity inside the MOF pores, and for introducing other functional groups onto their framework. The use of phosphine functions opens new possibilities for preparing porous materials with adjustable size of pores.
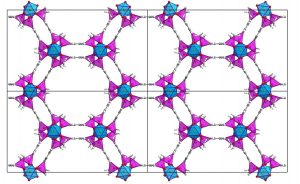
Structure of a phosphinate MOF ICR-2 (Inorganic Chemistry Res). The illustration shows octahedrally coordinated atoms of iron shown in blue colour, and phosphinate tetrahedrons in purple.
Hynek, J., Brázda, P., Rohlíček, J., Londesborough, M. G. S., Demel, J.*: Phosphinic Acid Based Linkers: Building Blocks in Metal-Organic Framework Chemistry. Angewandte Chemie – International Edition. 2018, 57, 5016-5019; JRC/D1.
Collaboration: Institute of Physics of the Czech Academy of Sciences