Our colleagues Josef Holub and Drahomír Hnyk from the Department of Syntheses in cooperation with scientists from the University of Pardubice, Institute of Organic Chemistry and Biochemistry of the ASCR, v.v.i. and from Auburn University (Alabama, USA) published an article in the prestigious journal Nature Communications that deals with the preparation and structural characterization of the first carborane cations in terms of the reactions of ten-vertex closo systems with N-heterocyclic carbenes followed by either the acidification with hydrogen chloride or the reaction with phosphorus trichloride. Geometrical arrangements of these cations comply both with the nido and closo electron counts.
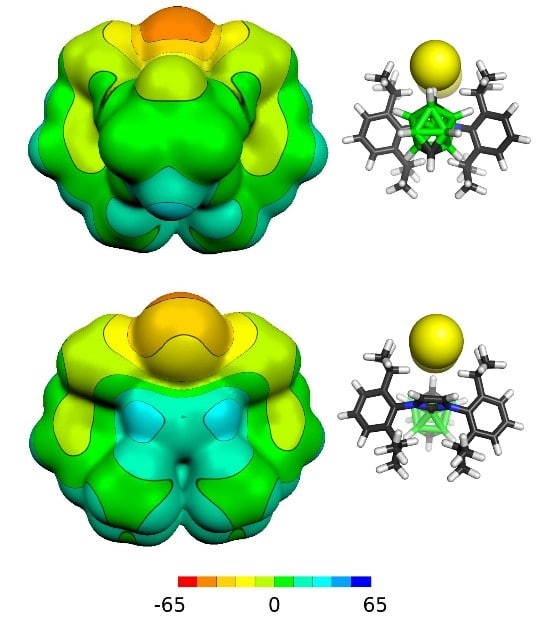
Calculated electrostatic potential of one of the experimentally verified cations with closo electron arrangement
The resulting cationic compounds are quite stable in the air and are soluble in water, which represents the principal difference in relation to the so far known neutral or anionic species, the later being very sensitive or even pyrophoric. These new materials can significantly contribute to such areas, where the transfer to surroundings is easier for the cations as a whole. This can be the case of either Proton Boron Capture Therapy (PBCT) that requires the presence of the 11B isotope in a cancer cell or in the areas of homogeneous catalysis or optoelectronic applications.
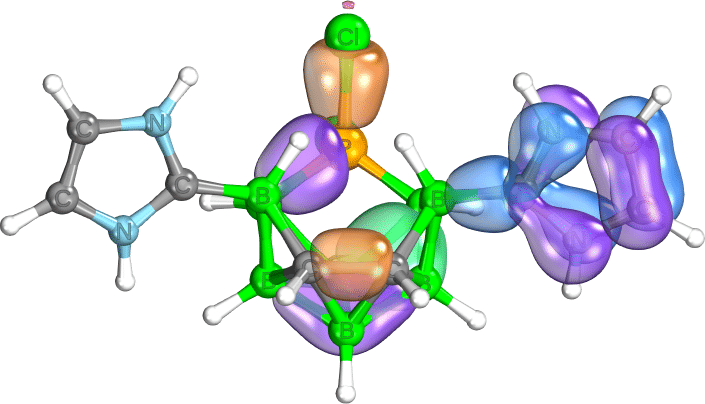
Bonding ratios of cation with nido electron arrangement
You can read the whole article here.